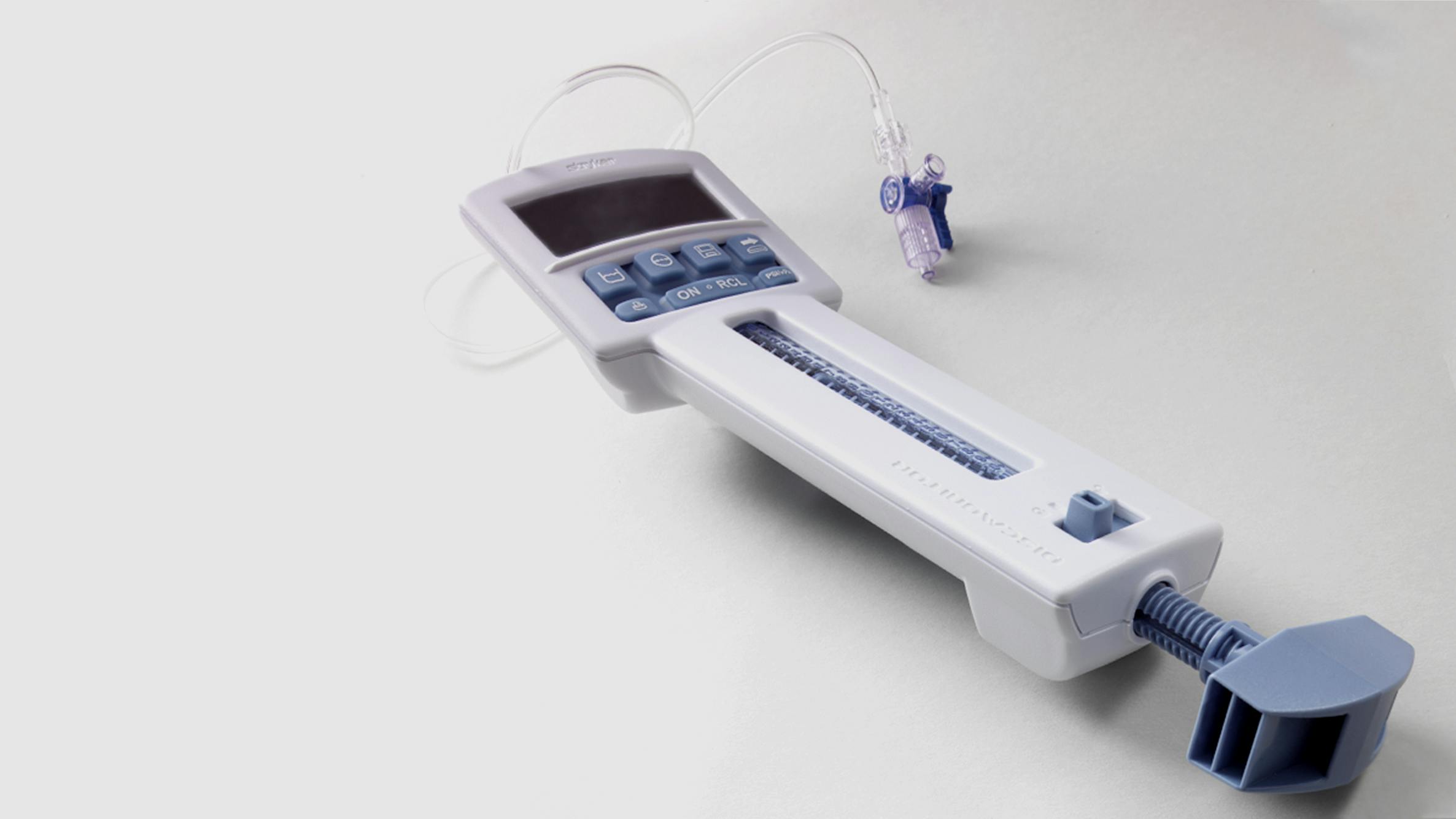
Discmonitor™
Stryker Instruments’ Interventional Pain Division approached us to design a disposable, sterile, digital discography tool — a minimally invasive diagnostic device to assess the extent of intervertebral disc damage and determine the source of back pain.
Device helps doctors diagnose damaged discs
Award
Winner
Winner
Otometrics: Otoscan
Laser scanning technology could create a 3D digital image of the complete ear. But to work, a headset placed around the patient’s ear during the scan had to be completely immobile.
Read more
ProNova: SC360 Proton Therapy System
Imagine being strapped into a large machine, completely immobilized, for an hour while it delivers high doses of radiation to treat your cancer - every day for six months.
Read more
Onelink® Smoke & CO Detector
First Alert engaged us to develop a fresh aesthetic that would communicate their latest technology in an appropriate premium-design statement.
Read more
Award
Winner
Winner
EXOGEN® Ultrasound Bone Healing System
EXOGEN® is the only FDA-approved bone healing device that uses ultrasound to stimulate the body’s natural healing process.
Read more